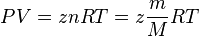Difference between revisions of "Darcy's law"
From wiki.pengtools.com
(Created page with "== Brief == Real Gas is a gas whose molecules occupy space and have interactions [https://en.wikipedia.org/wiki/Real_gas]. Real Gas deviates from Ideal Gas by pr...") |
|||
| Line 29: | Line 29: | ||
[[Category:E&P Portal]] | [[Category:E&P Portal]] | ||
| + | [[Category:pengtools]] | ||
Revision as of 06:12, 23 April 2018
Brief
Real Gas is a gas whose molecules occupy space and have interactions [1].
Real Gas deviates from Ideal Gas by proportionality term z, which is gas compressibility factor.
Equation Of State
Nomenclature
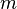 = mass, lbm
= mass, lbm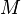 = molecular weight, lbm/lbmol
= molecular weight, lbm/lbmol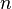 = number of moles, lbmol
= number of moles, lbmol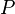 = Pressure, psia
= Pressure, psia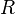 = universal gas constant, 10.7316 psia ft3/ lbmol/ °R
= universal gas constant, 10.7316 psia ft3/ lbmol/ °R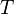 = temperature, °R
= temperature, °R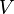 = volume, ft3
= volume, ft3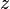 = gas compressibility factor, dimensionless
= gas compressibility factor, dimensionless